
Kobalt Oxide Pada ʻEleʻele Co3O4 Pauda
Hōʻike huahana
ʻO ka pauka Cobalt tetroxide he pauka ʻeleʻele, hiki ʻole ke hoʻoheheʻe ʻia i ka wai, me ka luster metala a me ka conductivity uila maikaʻi.ʻO ka Cobalt tetroxide he oxidizing nui a hiki ke hoʻokuʻu i ka oxygen i loko o kahi ʻakika.ʻO ka pauka Cobalt tetroxide he mea hoʻoikaika nui, hiki ke hoʻohana ʻia e synthesize i ka ammonia, formaldehyde, oxalic acid a me nā mea ʻē aʻe.ʻO ia hoʻi kahi mea magnetic kiʻekiʻe a me ka puaʻa.I ka hana ʻana i ka pākaukau, hoʻohana pinepine ʻia ia ma ke ʻano he mea electrode maikaʻi, hiki ke hōʻemi pono i ka pale kūloko o ka pā.Eia kekahi, hiki ke hoʻohana ʻia ma ke ʻano he precursor no nā lubricants a me nā mea superconducting kiʻekiʻe.
Hōʻike
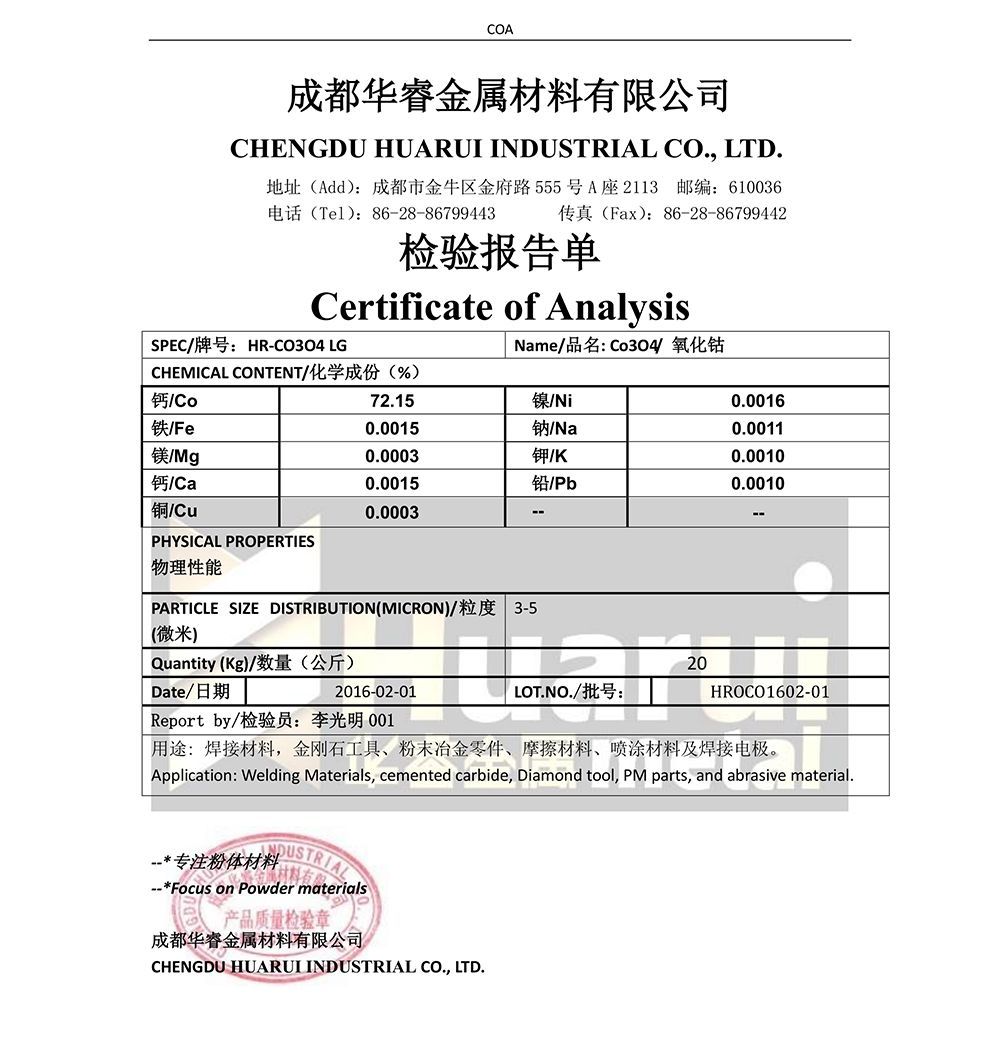
| ʻO ka hoʻohui ʻana i ka pauka Cobalt oxide | ||||||
| Papa | Ka haumia i loko (wt% max) | |||||
| Co% | Ni% | Cu% | Mn% | Zn% | Fe% | |
| A | 73.5±0.5 | ≤0.05 | ≤0.003 | ≤0.005 | ≤0.005 | ≤0.01 |
| B | ≥74.0 | ≤0.05 | ≤0.05 | ≤0.05 | ≤0.05 | ≤0.1 |
| C | ≥72.0 | ≤0.15 | ≤0.10 | ≤0.10 | ≤0.10 | ≤0.2 |
ʻO Coa
Palapala noi
1. Hoʻohana ʻia e like me ka waihoʻoluʻu a me ka puaʻa no ke aniani a me nā seramika, ka paʻa paʻa;
2. ʻO nā oxidants a me nā catalysts i ka ʻoihana kemika;
3. Hoʻohana ʻia i ka ʻoihana semiconductor, nā mea uila uila, lithium ion battery cathode material, magnetic materials, temperature and gas sensors;
4. Hoʻohana ʻia e like me ke kiʻekiʻe maʻemaʻe analytical reagent, cobalt oxide a me ka hoʻomākaukau paʻakai cobalt
Pūnaehana hoʻomalu maikaʻi

He ʻōnaehana koʻikoʻi ko Huarui.Ho'āʻo mua mākou i kā mākou huahana ma hope o ka hoʻopau ʻana i kā mākou hana, a hoʻāʻo hou mākou ma mua o kēlā me kēia lawe ʻana, ʻo ia hoʻi ka laʻana.A inā makemake ʻoe, makemake mākou e ʻae i ka ʻaoʻao ʻekolu e hoʻāʻo.ʻOiaʻiʻo inā makemake ʻoe, hiki iā mākou ke hāʻawi i kahi laʻana iā ʻoe e hoʻāʻo.
Hoʻopaʻa ʻia kā mākou huahana huahana e Sichuan Metallurgical Institute a me Guangzhou Institute of Metal Research.Hiki i ka hui lōʻihi me lākou ke mālama i ka manawa hoʻāʻo nui no nā mea kūʻai aku.











